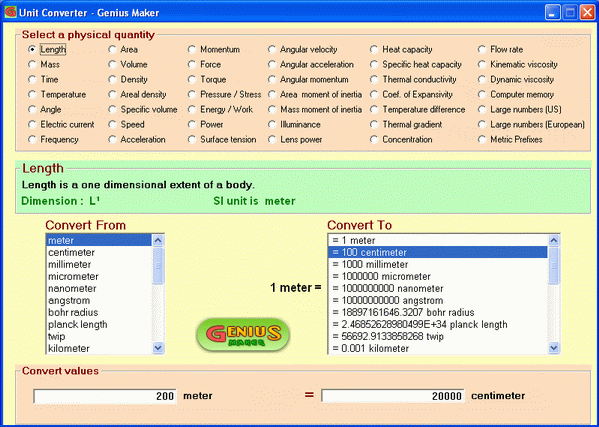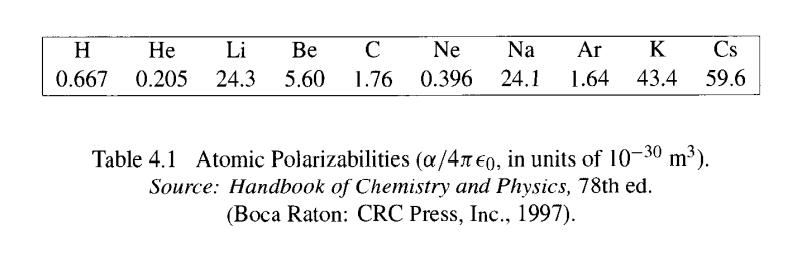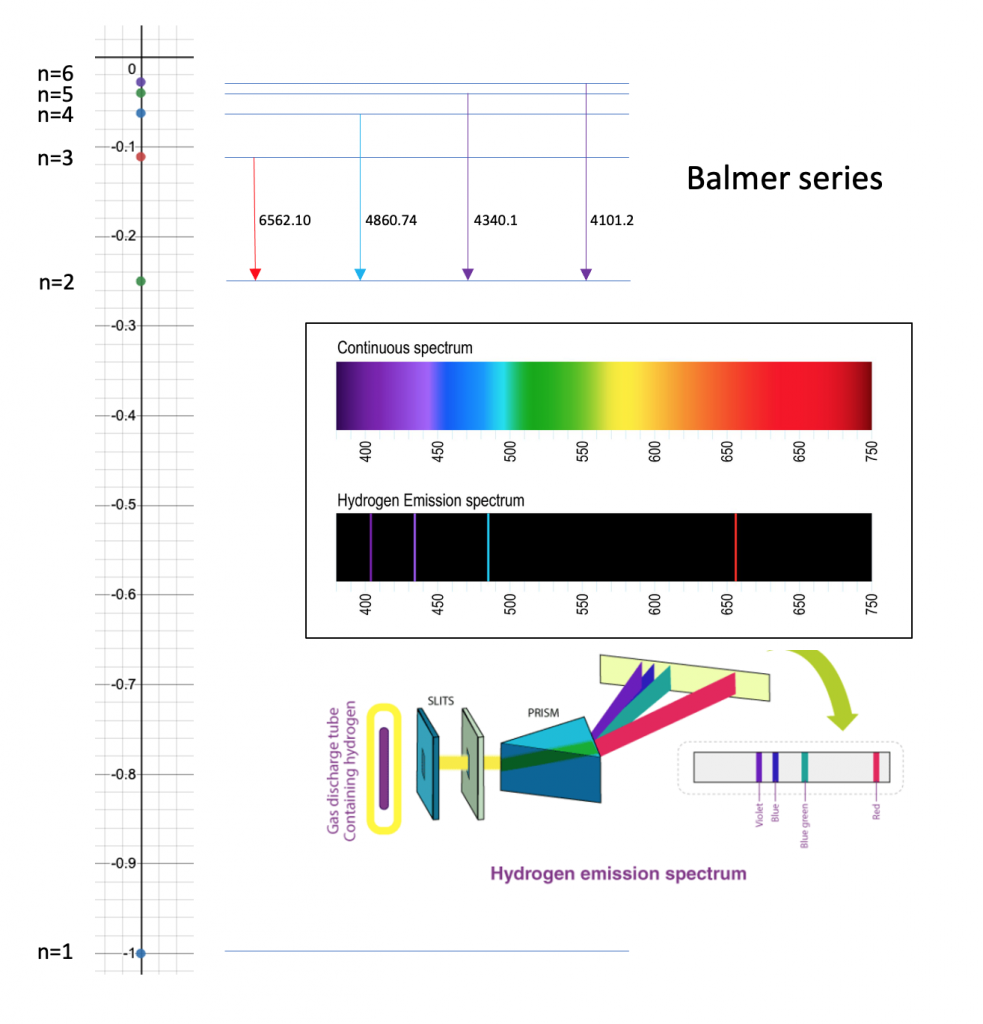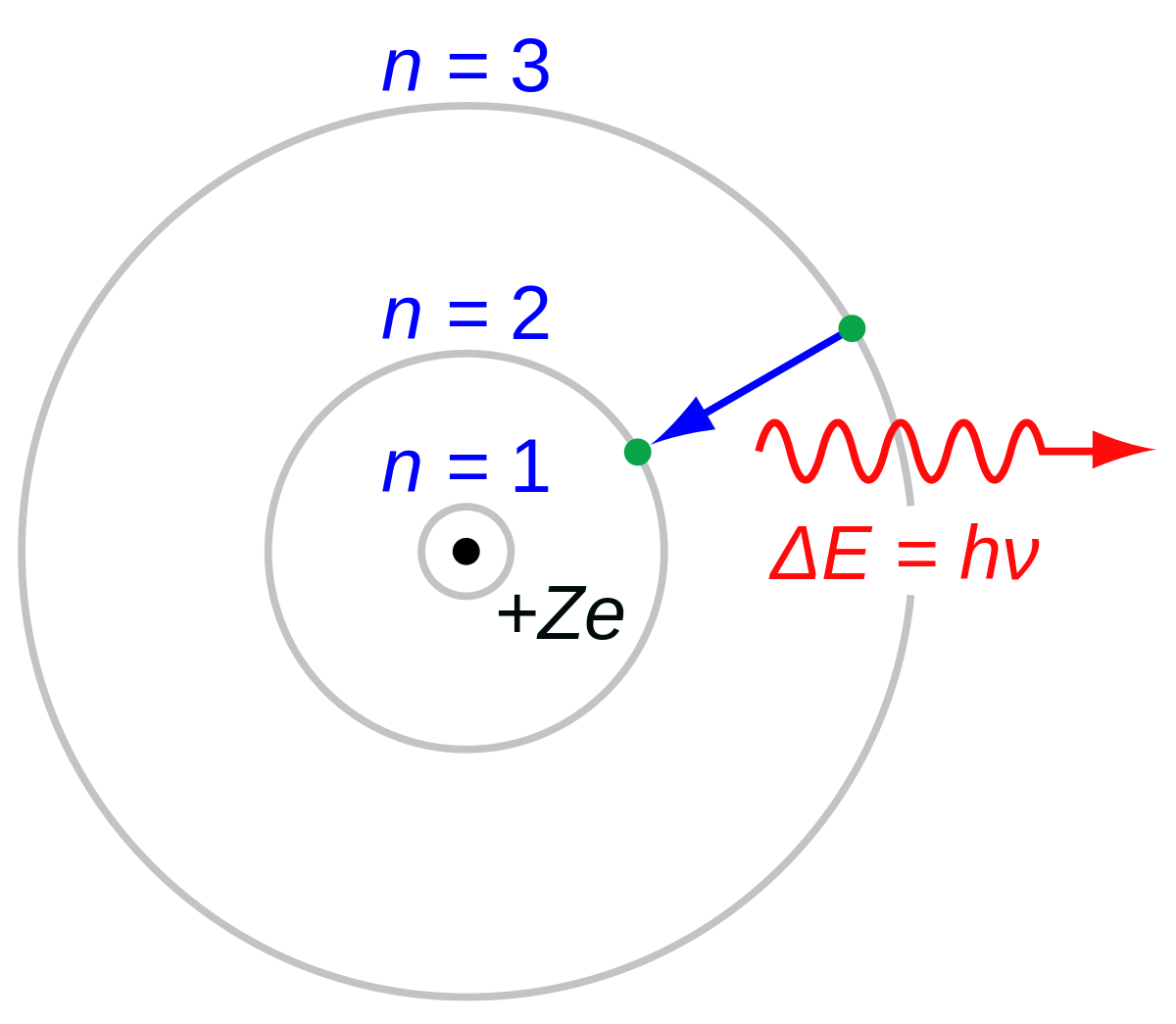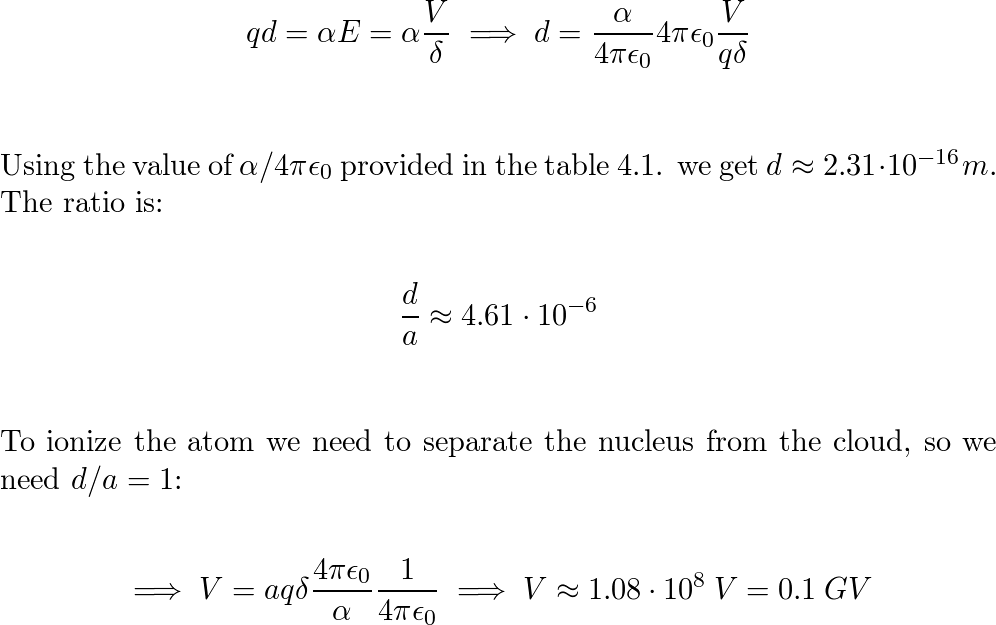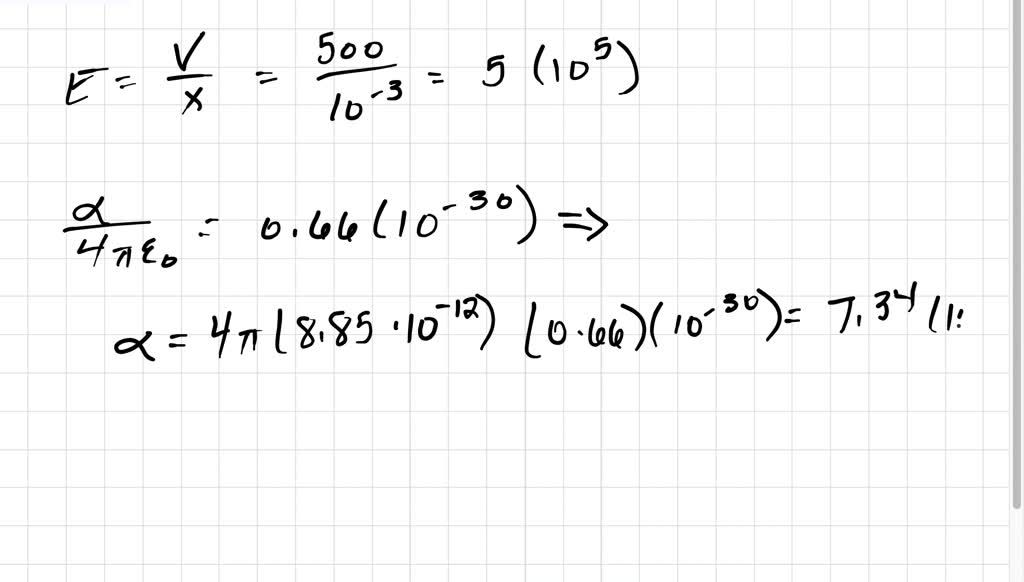
SOLVED:A hydrogen atom (with the Bohr radius of half an angstrom) is situated between two metal plates 1 mm apart, which are connected to opposite terminals of a 500 V battery. What

the radius of first bohr Orbit for hydrogen is 0.53 Amsterdam the radius of third Bohr's orbit will - Brainly.in

Calculate the wavelength in angstrom of the photon that is emitted when an electron in Bohr orbit n = 2 returns to the orbit n = 1 in the hydrogen atom. The

Calculate the wavelength in Angstroms of the photon that is emitted when an electron is Bohr orb... - YouTube

Bohr Model of the Atom. Experimental Observation of Hydrogen Line Emission In 1853, Anders Angstrom of Sweden first determined that a set of discrete. - ppt download

Bohr radius for hydrogen atom ( n=1) is approximately 0.530 angstroms. What is th radius for first excited - Brainly.in

WARNING in particle_methods.F:684 :: The distance between the atoms *** *** 10 and 405 is only 0.496 angstrom and thus smaller than the threshold *** *** of 0.500 angstrom
Calculate the wavelength in Angstroms of the photon that is emitted when an electron in the Bohr's orbit, n = 2 returns to the orbit, - Sarthaks eConnect | Largest Online Education Community

SOLVED:(a) According to the Bohr model, an electron in the ground state of a hydrogen atom orbits the nucleus at a specific radius of 0.53 Å. In the quantum mechanical description of