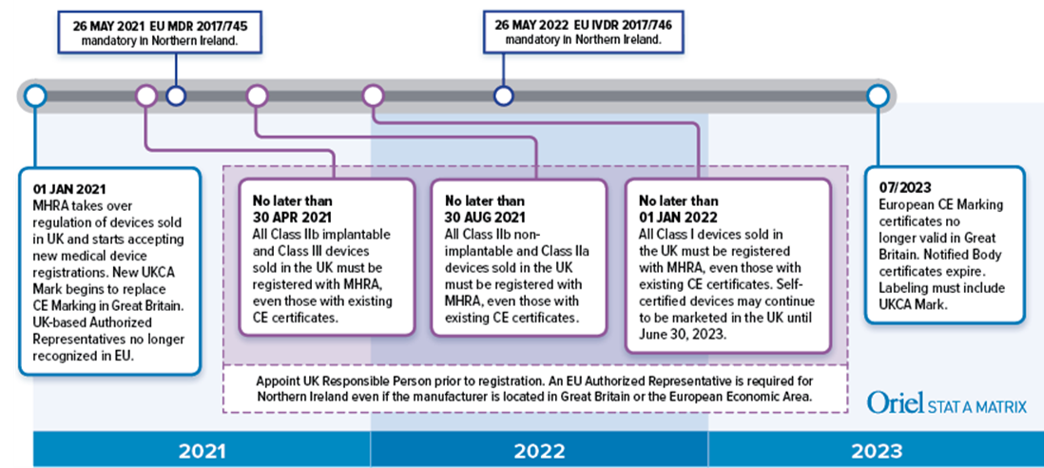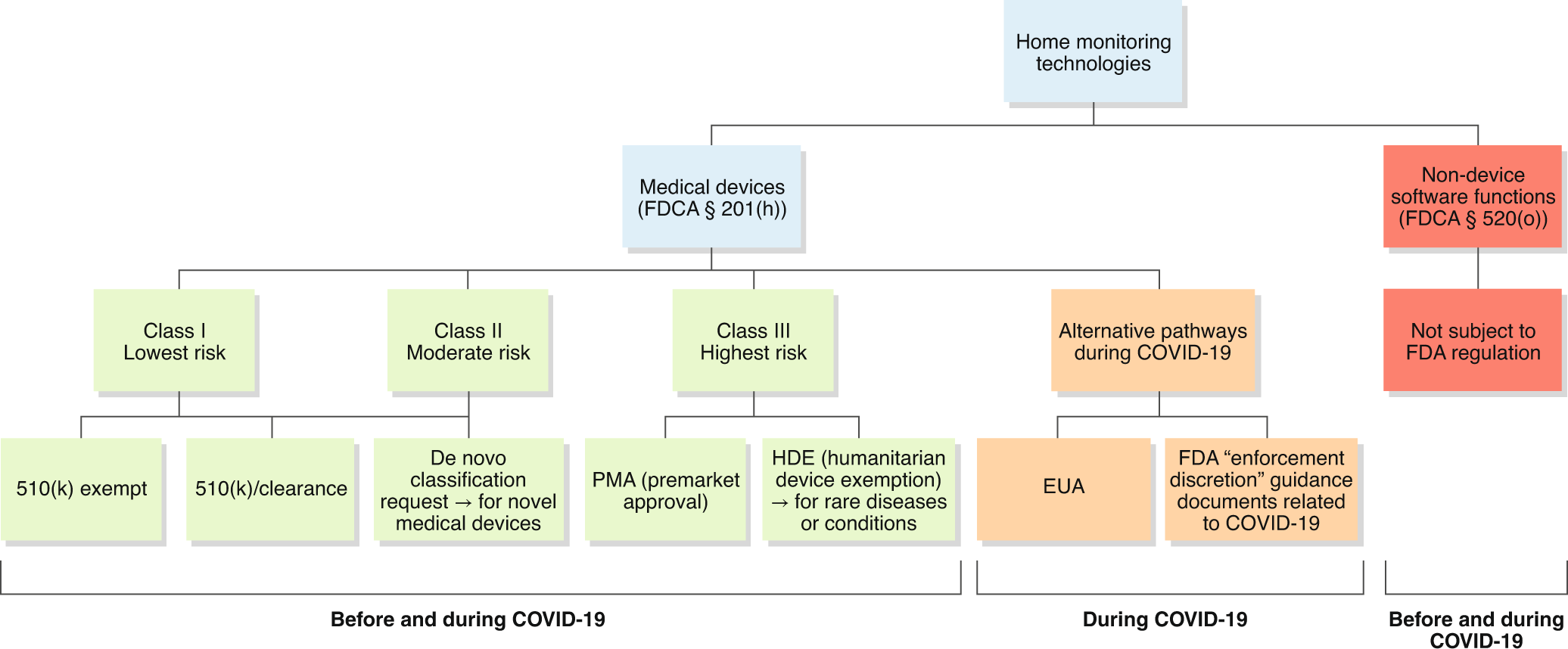
Regulatory, safety, and privacy concerns of home monitoring technologies during COVID-19 | Nature Medicine

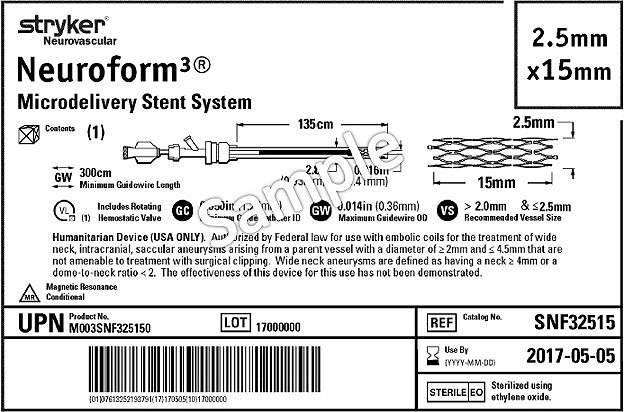
Guidance on Label and Instructions for Use for Medical Devices - FDA Regulatory Consulting and Training Services

ISO 15223-1:2021(en), Medical devices — Symbols to be used with information to be supplied by the manufacturer — Part 1: General requirements
PLOS Neglected Tropical Diseases: The Art of Writing and Implementing Standard Operating Procedures (SOPs) for Laboratories in Low-Resource Settings: Review of Guidelines and Best Practices